Numeta G13E
Numeta G13E is available in select European countries; it is not approved for use in the United States.
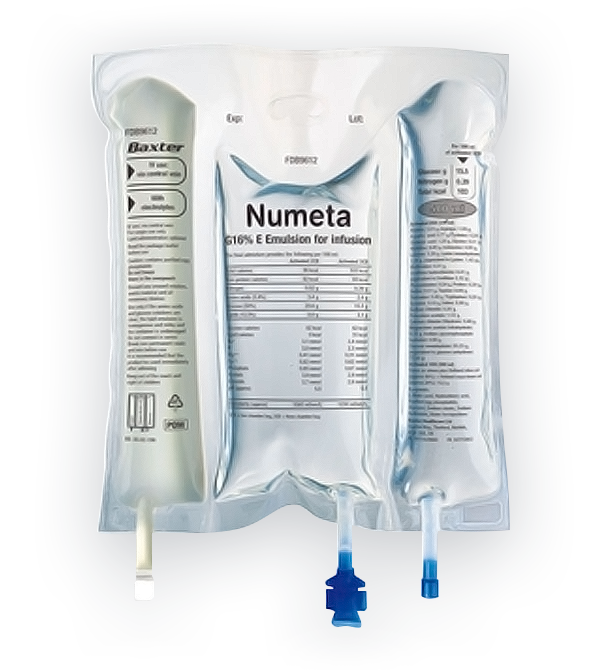
Numeta G13E 300 mL is the only triple-chamber, ready-to-use parenteral (intravenous) nutrition (PN) product available to treat preterm infants (less than 37 weeks gestation age) who are at high risk for infection and malnutrition5 in the early hours and days of their lives.
Numeta G13E is formulated to follow the European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) and The European Society of Clinical Nutrition and Metabolism (ESPEN) guidelines to meet neonatal nutritional needs with a balanced formulation of amino acids (protein), glucose (carbohydrates), lipids (fats) and electrolytes.
Numeta G13E is designed for activation and administration at the bedside; research indicates ready-to-use PN may reduce the potential risk of medication errors and associated infections.1
Numeta is also available in additional formulations, including Numeta G16E 500mL for full term infants through two years of age, and Numeta G19E 1,000mL for children and adolescents (2-18 years of age).
Product Details
Flexible
Numeta is flexible to meet patient needs day by day
Activate the lipid chamber only as needed 2-4
Add other ingredients as needed 2-4
Timely Nutrition Support
Can be stored at room temperature 2-4
Longer shelf-life than compounded PN (18-24 months) 2-4