Oxiris
The Oxiris set is available in select countries around the world.
Oxiris is the only blood purification filter set to effectively remove inflammatory mediators, endotoxin, fluid, and uremic toxins simultaneously. 1, 2
Innovative Technology
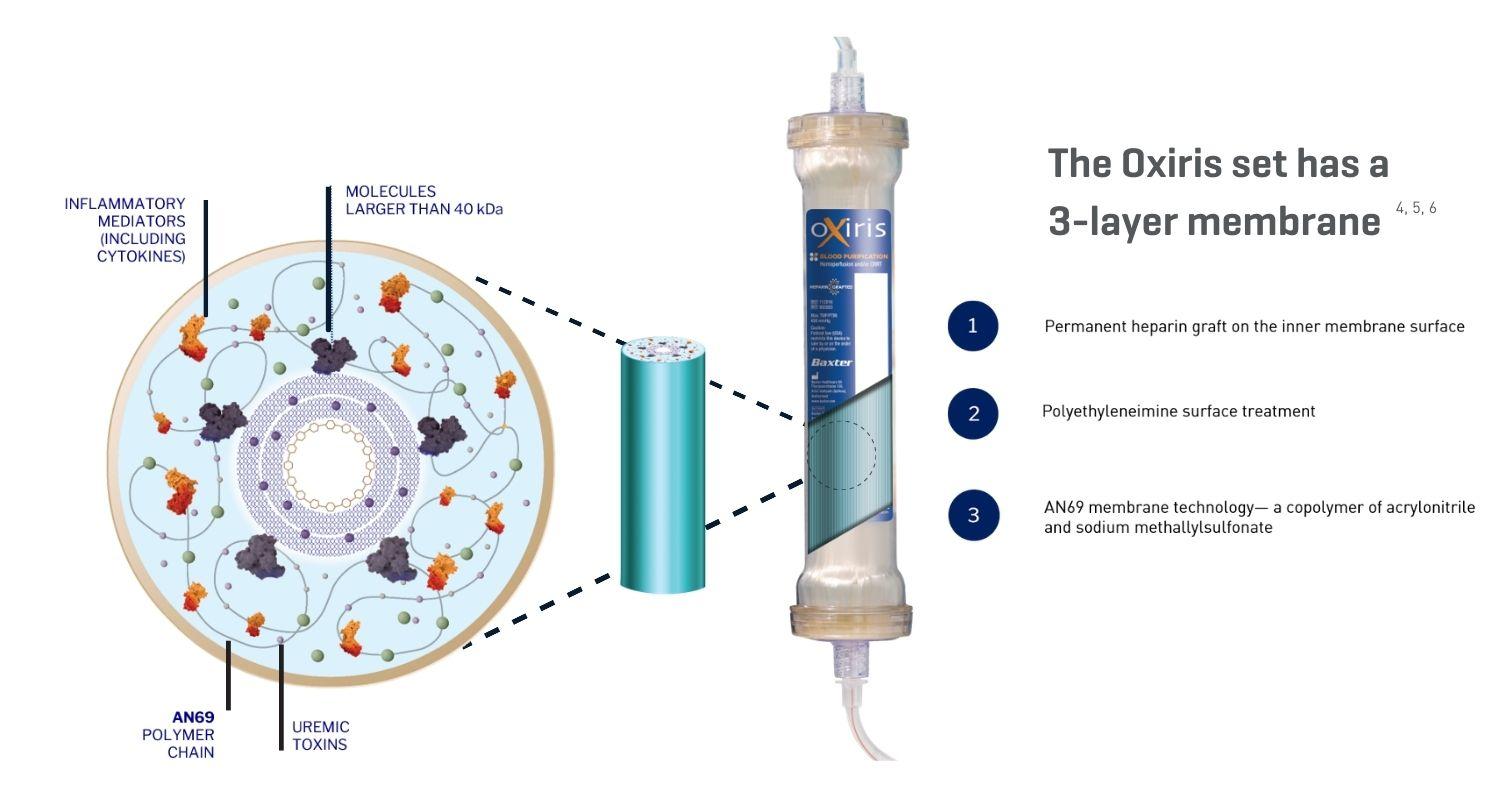
The Oxiris set membrane delivers functional benefits3:
Product Features
The Oxiris set is designed to simplify blood purification therapy delivery for patients requiring removal both of inflammatory mediators/endotoxin and CRRT simultaneously
Versatile Innovation
The Oxiris set can be used to deliver hemoperfusion to reduce elevated levels of inflammatory mediators/endotoxin and/or any CRRT modality (SCUF, CVVH, CVVHD, CVVHDF) to provide uremic toxin removal and support fluid management.3
CVVH, Continuous Veno-Venous Hemofiltration; CVVHD, Continous Veno-Venous HemoDialysis; CVVHDF, Continuous Veno-Venous HemoDiaFiltration; SCUF, Slow Continuous UltraFiltration
Versatile Blood Purification
- Inflammatory Mediator Removal: A prospective, multicenter, observational study reported significantly decreased serum IL-6 levels from baseline in patients with COVID-19 treated with the Oxiris set.2
- Endotoxin Removal: A randomized, crossover study reported significantly decreased inflammatory mediator levels from baseline, specifically IL-6, IL-8, and endotoxin, in patients with septic shock treated with the Oxiris set.1
Versatile Simplification
- The Oxiris set is designed to simplify blood purification therapy for patients requiring removal of both inflammatory mediators and endotoxin.3
- The Oxiris set is part of a pre-connected set for simplicity. The closed system includes a filter membrane pre-connected to color-coded tubing, minimizing set-up steps and reducing potential for touch contamination.3, 7
Indications
The Oxiris set is indicated for use in critically ill patients with a body weight equal or greater than 30 kg (66lb) for hemoperfusion and / or renal replacement modalities such as:
- Slow Continuous UltraFiltration (SCUF)
- Continuous Veno-Venous Hemofiltration (CVVH)
- Continuous Veno-Venous HemoDialysis (CVVHD)
- Continuous Veno-Venous HemoDiaFiltration (CVVHDF)
When used for hemoperfusion only, the SCUF mode shall be used with no fluid removal prescription, as the indication is to reduce elevated levels of inflammatory mediators, such as cytokines, and to reduce endotoxins.
If patients suffer from acute kidney injury and / or volume overload, the Oxiris set is indicated for continuous renal replacement therapies (CRRT), to perform fluid management and removal of uremic toxins. The removal of inflammatory mediators and endotoxins is performed simultaneously when indicated for CRRT.
Intended Purpose
The Oxiris set is a single use device that provides blood purification via diffusion, convection and adsorption through a semipermeable membrane.
The Oxiris set is for use only in conjunction with the Prismaflex control unit or with the PrisMax control unit (in countries where PrisMax is cleared or registered).
All treatments administered with the Oxiris set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
Important Risk Information
The use of Oxiris set is contraindicated (as mentioned in the IFU) when:
- Patients present a known allergy to heparin or have type II thrombocytopenia caused by heparin (HIT Syndrome type II)
- A drug used simultaneously with Oxiris set is contraindicated per its Instructions for use
Relative contraindications (individual risk/benefit to be determined by treating physician) for the use of Oxiris include:
- The inability to establish vascular access to safely perform CRRT / hemoperfusion (SCUF; CVVH; CVVHD; CVVHDF)
- Severe hemodynamic instability
- Known hypersensitivity to any component of the Oxiris Set
This set is intended for use in the following veno-venous therapies: SCUF; CVVH; CVVHD; CVVHDF.
All treatments administered with the Oxiris set must be prescribed by a physician. The size, weight, state of uremia, cardiac status, and general physical condition of the patient must be carefully evaluated by the prescribing physician before each treatment.
For safe and proper use of the products mentioned herein, please refer to the Operator's Manual or Instructions for Use.
 |